Multiple Factor Disease “Kennel Cough”
The very common airway disease, known as “Infectious Canine Laryngo-tracheitis” or „Kennel Cough“ is characterized by acute onset of an extreme rough, sometimes convulsive cough. A multi-factor disease is your first suspicion, involving both viral and bacterial pathogens. Predisposing factors are high population density, as can be seen in shelters, kennels or large breeding facilities. Here is, as at dog schools or dog pensions, a very high infection pressure. Animals at all ages can be affected, although puppies between 6 weeks and 6 months are affected the most and most severely ill.
On top of the list of pathogens are the viral once, such as Canine Adeno virus Type 2 (CAV-2) and Canine Parainfluenza virus (CPiV). An infection with CAV-1, the pathogen of Hepatitis Contagiosa Canis (HCC) can in mild cases also cause respiratory symptoms. More rarely are the Canine Influenza virus, Canine Distemper virus (CDV) or Canine Herpes virus (CHV) involved in the disease. The primary bacterial pathogen is the Bordetella bronchiseptica. After the respiratory epithelia cell damage caused by the above mentioned pathogens, secondary bacteria or mycoplasma infections can create more severe respiratory disease. As secondary pathogens Pseudomonas spp., E.coli, Klebsiella, Pasteurella and Streptococci can be seen.
In uncomplicated cases a dry or cramping cough is the only symptom seen, which can be stress-induced or induced by pressure on the larynx. The trachea is easily triggered. More severe cases are presented with anorexia, moist productive cough, lethargy, dyspnoea, tachypnoea and/or weakness. Fever is usually only moderate if present.
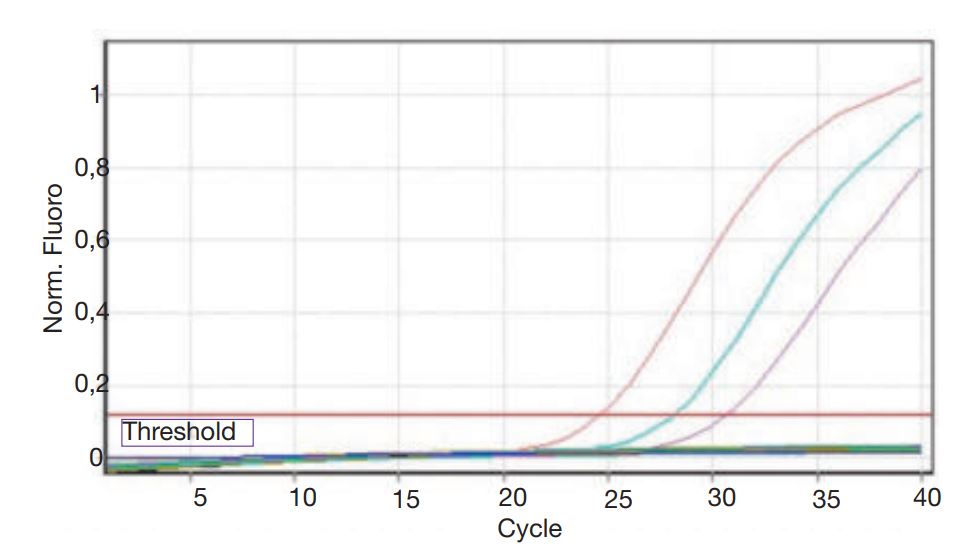
- Fig. 1: Real-time-PCR fluorescence curve: By an increasing curve the pathogen was demonstrated to be present in the sample material.
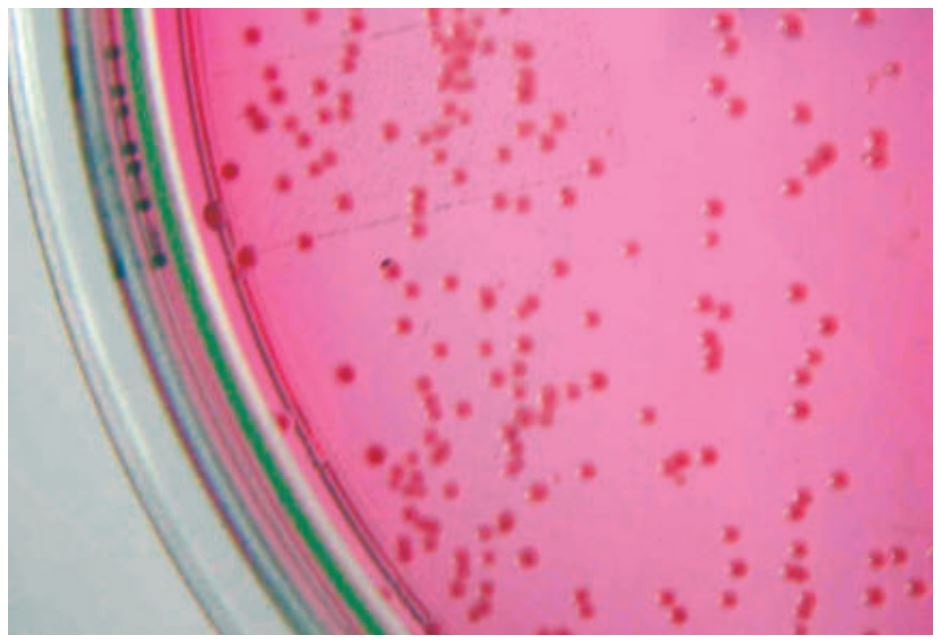
- Fig. 2: Bordetella bronchiseptica – Single culture on a selective culture medium
Correct diagnose is made from the history (sudden onset of symptoms, possible exposure (contact to other dogs), lack of immunization, etc.) combined with the clinical symptoms. Viral pathogens can, especially in the early phase, be detected by means of PCR from a nasal- or throat swab. As bacterial infections are the cause of the more severe symptoms, a bacteriology examination including culture and subsequently antibiogram is crucial. A culture swab taken deep in the throat or from a tracheal- or broncho-alveolar lavage (BAL) is needed. Sampling must be done before any antibiotics are given. (Fig. 1)
Antibody detection is not always as useful due to the huge distribution of these pathogens in the dog population, and one cannot distinguish between a vaccination or infection titre. A second antibody titre test after 2-3 weeks is therefore required to indicate a current infection in most cases.
Therapy consists in most cases of antibiotics according to the antibiogram for a minimum of 14 days. Cough medicine (e.g. Butorphanole) should only be given to dry coughs. Important is strict rest for at least 2-3 weeks; in severe cases even longer. The owner should be informed that their dogs is contagious and should be kept away from other dogs. Especially by Bordetella bronchiseptica infections as it can persists in the respiratory tract for several months after recovery. As prophylaxes several vaccines are available.
Canine Parainfluenza virus (CPiV)
CPiV is a large enveloped RNA-Virus belonging to the Paramyxo virus family. It cannot replicate in macrophages and is therefore only to be found in the upper respiratory system. Mono-infections run rather unapparent but can be with respiratory symptoms like nasal discharge or cough.
Shedding of virus occurs mostly in the early stage of infection (6-8 days p.i.) in oro-nasal secretions. Detection is possible by PCR test.
Canines Adeno virus (CAV)
CAV is a non-enveloped DNA-virus with relative high tenacity. Adeno viruses are strictly species specific. CAV-1 and CAV-2 are genetically and anti-genetically close related; their antibodies show cross protectivity. Their difference lies in their organ tropism. CAV-2 show affinity for the respiratory tract: the virus replicates in the ciliated epithelium. Opposite the CAV-1, CAV-2 does not spread into a generalized infection. The clinical symptoms are often mild.
CAV-2 are excreted in nasal secrete up to approximately 9 days p.i. Contamination occurs through aerosols and direct contact. Detection is possible by PCR test.
Canine Distemper virus (CDV)
Canine Distemper virus (CDV) is a high-contagious RNA-Virus which affects several species in the order Carnivora. Besides dogs also fox, ferret, raccoon, otter and more can be infected. It causes acute to chronic general disease with different symptoms depending on the immune status of the animal:
- GI-symptoms: Diarrhoea, vomitus
- Respiratory symptoms: Conjunctivitis, coughing, bronchitis through to catarrhal broncho-pneumonia
- CNS-symptoms: Blindness, ataxia, behavior changes, tonical-clonical cramps
- Cutaneous symptoms: Hyperkeratosis of the feet or nares
Virus reservoir is clinical unapparent infected animals. Contamination occurs through direct contact or by aerosols. The initial virus replication occurs in the mono-nucleated cells in the local lymph nodes or tonsils wherefrom it evolves to a cell-associated viremia and target organ infection.
Young dogs from 6 months age are most susceptible. Typical symptoms are intermittent fiver. Virus are excreted with every kind of body secrete from day 5 p.i. The direct detection for diagnosis is possible by PCR test.
Canine Herpes virus (CHV)
Canine Herpes virus (CHV) infection is a very per-acute disease in young puppies; it can cause abortion if the infection is intrauterine, but CHV is in adult animals mostly an unapparent infection with only mild respiratory symptoms like rhinitis and conjunctivitis, if any.
Herpes virus is a large enveloped DNA-virus which is highly host-specific. Typical herpes virus infections are life-long latent infections. In the latent phase virus is situated in the neurons of the sensory ganglia. A sporadic reactivation of a latent infection can occur due to stress or immune suppression (e.g. Glucocorticoids), whereby CHV are shed and can be found in oro-nasal and vaginal secretions.
Contamination occur mostly oro-nasal through direct contact with an infected animal as intrauterine. Thereafter virus replication occurs in the mucous membrane of the upper respiratory tract and in the tonsils. Virus replication occurs at an optimum temperature of 37°C. Therefore, in puppies with a reduced body temperature, a viremia and subsequently a haemorrhagic-necrotizing infection of the inner organs occur. In older puppies where the body temperature regulation is fully functional the infection is limited to the upper respiratory tract. Virus detection by PCR test of nasal- or throat swab can be useful in active phases of infection. Alternatively PCR test on liver tissue upon autopsy or uterus swab from the aborting bitch.
Bordetella bronchiseptica
Bordetella bronchiseptica is a small, aerobe and mobile gram-negative bacteria. (Fig. 2)
It has a high affinity for ciliated epithelium in the respiratory tract and produces a row of toxins which paralyzes the cilia, inhibits the mucociliary clearance and decreases the phagocytosis. That is the reason why Bordetella bronchiseptica, besides it primary pathogenicity, plays an important role in facilitating secondary bacterial infections. It is multi-hostel (Zoonose) and can be contagious from dogs to cats and exotics and vice-versa.
Contamination occurs through droplet infection and close contact. The incubation time is 2-14 days. Clinical symptoms are predominately coughing and serous nasal- or eye secretion but otherwise unaffected.
Bordetella bronchiseptica causes a strong local immune response but as a facultative intracellular organism, it can escape the specific immune response and persist in the respiratory tract for a long period of time. Dogs which have been infected with Bordetella bronchiseptica have immunity for at least 6 months thereafter.
Detection of Bordetella bronchiseptica is mostly done by means of a PCR profile, where you profit from the extreme high sensitivity of the test method. A culture with subsequent antibiogram is recommended for correct therapy, as Bordetella bronchiseptica resistance to Doxycycline and Tetracycline has been described. Therapy should be continued until at least 1 week end of symptoms. As prophylaxes an intranasal life vaccine is available. After vaccination the bacteria enters the respiratory tract for several month and causes a local immunity. A vaccination during antibiotic therapy is therefore worthless.
Mycoplasma spp.
Mycoplasmas are very small organism without a cell wall which replicates independently through mitosis. Mycoplasmas require direct contact with host cells in order to have a functional metabolism. Therefore a special culture medium is needed for both transportation and cultivation and the organism also grow much slower than bacteria in the lab. Mycoplasmas are often not very virulent, but on the other hand very capable of avoiding the infected animal’s immune response. Infections are often secondary to other diseases. Pathogenic Mycoplasmas show a degree of host specificity.
Mycoplasma-commensal can be found in the canine naso-pharynx mucous membrane but is normally not to be found in the lower respiratory tract. Here they can cause cilia stasis, loss of cilia or even purulent bronchopneumonia if the lower respiratory tract has been pre-damaged by e.g. virus. Especially Mycoplasma cynos can be found in conjunction with respiratory disease in dogs.
Diagnostic samples should be taken from the lower respiratory tract. PCR test can be performed directly from the BAL-sample or from a dry swab.
Due to lack of a cell wall, only antibiotics impacting the protein- or nuclear acid syntheses are effective, as gyrase-inhibitors, macrolides- or tetracycline antibiotics. Please notice that therapy must be carried out for at least 4-6 weeks.
Streptococcus equi subsp. zooepidemicus
During the past years more and more reports of acute haemorrhagic pneumonia have been reported in dogs. Mostly dogs in large groups are affected (e.g. kennels, shelters). Responsible pathogen was identified as being Streptococcus equi subsp. zooepidemicus. Some were co-infections with the above described virus which also here can act as a precursor for secondary bacterial infections.
Initial the dogs show only nasal discharge and moist coughing, but later tachypnoea, anorexia and fiver sets in. Sometimes even a massive dyspnoea occur due to a haemorrhagic pleural effusion and death will follow within 2 days after first clinical symptoms are seen; mortality rate is high despite intensive therapy. The exact pathogenesis is still unclear.
The most suited material for culture or PCR tests are tracheal- or broncho-alveolar lavage (BAL) or lung tissue from death animals. As Streptococcus equi subsp. zooepidemicus is shed through respiratory secretes also nasal- or throat swabs can be used for PCR. Sick animals must in all cases be kept in isolation to avoid infecting other dogs.
Therapy should be initiated as soon as possible. Drugs of choice are Cephalosporin-, ß-Lactam- or Macrolide-antibiotics with a guarded prognosis.
Besides the above mentioned pathogens several mycological or parasitic infections can occasional play a role in canine respiratory disease.
Summery
For the diagnosis of canine infectious diseases causing coughing, especially in young animals with contact to other dogs, the following laboratory tests are significant:
• PCR Test:
Especially valuable in detecting virus, but useful for many pathogen bacteria as well. Please notice that no antibiogram is available after a PCR test.
Material needed: Tracheal- or BAL-sample, swab without medium or in rare cases a tissue sample.
• Bacteriology culture:
Culture with subsequent antibiogram for a correct therapy. Every pathogen found in a BAL-sample is usually relevant.
Material needed: Tracheal- or BAL-sample or swab with transport medium