Over the last few decades the popularity of reptiles as pets has increased. This trend is clearly reflected in the increasing number of exotic trade shows and the availability of reptiles in many pet shops. The animals are offered at low prices or even as an „all-inclusive set“ and purchased by unknowing pet lovers. Frequently the rude awakenings follow immediately – the reptiles become ill and are presented at the veterinary practice more and more often.
This of course provides a welcome variety in everyday practice life, but it also poses a new challenge to the practitioner.
There are some difficulties arising from differences between reptiles and the common mammals: the physiology of the ectothermic animals, their reaction to medications, available laboratory tests and last but not least a different morphology of blood cells.
Blood sampling
The blood sampling technique varies depending on the patient species (Tab. 1).Techniques such as clipping of toenails or tail tips are rendered obsolete and are no alternative to professional venipuncture.
General guidelines:
– A blood volume of 0.5% up to 0.8% of the body weight can be sampled safely from a healthy, normally hydrated animal
– The anticoagulant of choice is Li-Heparin, as EDTA may cause haemolysis, especially in tortoises and turtles
– Immediate preparation of a blood smear in order to avoid pre-analytical errors like the deterioration or clumping of blood cells
(Tab. 1)
Sample analysisk
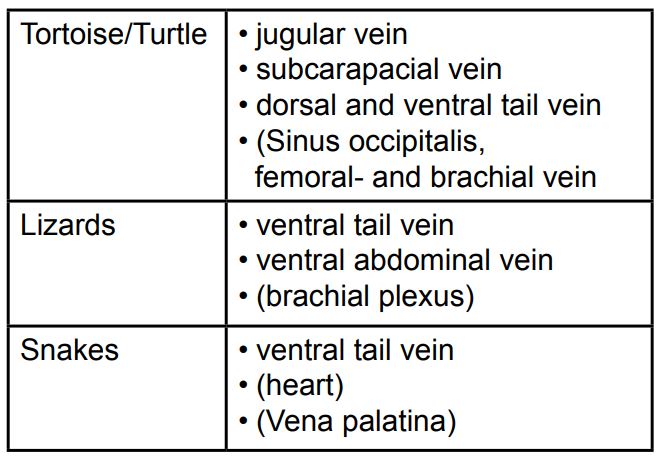
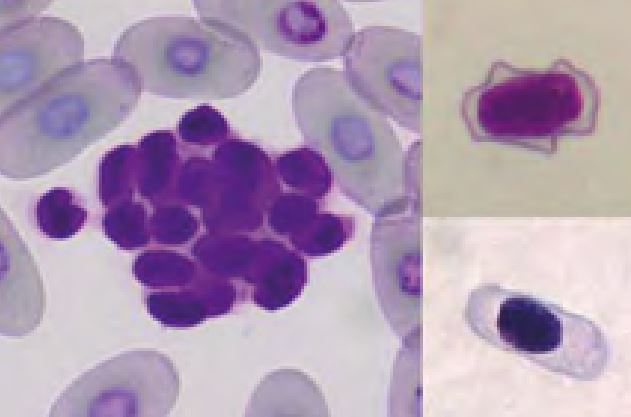
There is a fundamental difference between mammalian and reptilian blood cells: In reptiles all blood cells are nucleated, whereas in mammals only leukocytes and erythroid precursors show a nucleus (Fig.2).
In general, haematology analysers first count In general, haematology analysers first count erythrocytes and thrombocytes, then lyse these cells and subsequently count the nucleated leukocytes. Unfortunately the nuclei of reptilian erythrocytes and thrombocytes do not lyse and therefore a reliable and exact separation of the different cell lines is not possible. Hence automated cell counters are not applicable for reptile blood samples.
Blood cell counts are performed by manual methods:
One possibility is the use of a haemocytometer with different staining and solution systems (e.g. the Unopette system or Natt and Herrick’s solution). These techniques are time consuming and results actually show a higher variability than results of automated methods.
Another manual technique is the estimation of leukocytes and thrombocytes using a stained blood smear. With this method the variability of results is also high, but the procedure is rapid and simply performed with a little bit of experience. Using the 40x objective (400x magnification, if required 100x objective/ 1000x magnification) leukocytes are counted in at least 10 fields of the blood smear. The average cell count per field is calculated and multiplied by the square of the magnification of the used objective (i.e. with 400x magnification by factor 1600, with 1000x magnification by factor 10000). The result is the number of cells per μl blood.
A small calculation example:
7 Leukocytes/400x field
7×1600=11200
The leukocyte count is 11200/μl
Thrombocytes are counted in the same way with the 100x objective (1000x magnification) but multiplied by factor 15000.
Furthermore the differential leukocyte count and morphology of the blood cells are obtained by microscopic examination of blood smears.
It is not possible to estimate the erythrocyte number. The packed cell volume (PCV) is determined by microhaematocrit centrifugation as in mammals. Haemoglobin concentration can be measured by haematology analysers.
Blood Cell Morphology
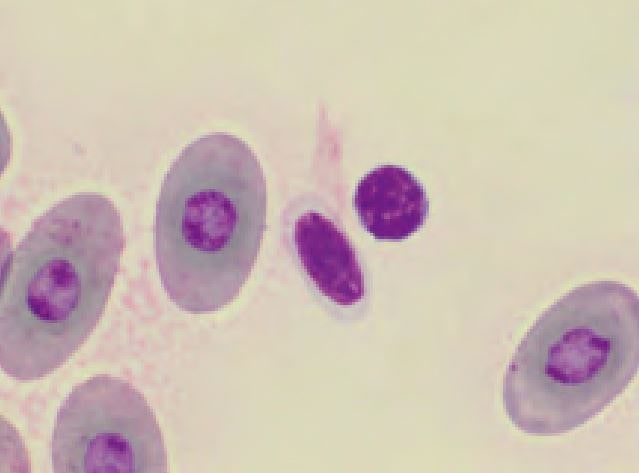
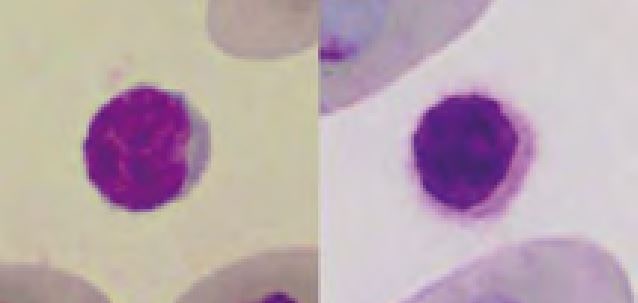
Erythrocytes of reptiles are ellipsoidal and have a centrally positioned nucleus. Depending on the stain used, the colour of the cytoplasm ranges from orange to pink or pale violet. Round basophilic or irregular clear inclusions (Fig. 3) are regularly found in the otherwise homogenous cytoplasm. In general these findings represent artefacts of slide preparation, staining or a prolonged drying period. Slight anisocytosis and polychromasia are normal in most reptiles. Increased numbers of polychromatic (juvenile) erythrocytes are found in regenerative anaemia, during ecdysis and in young animals.
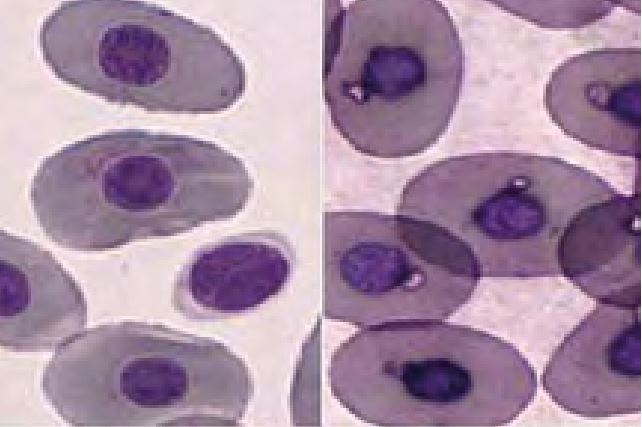
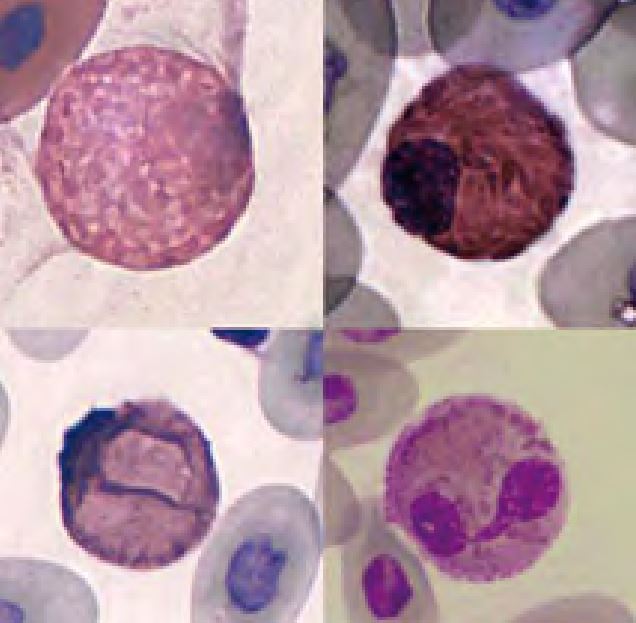
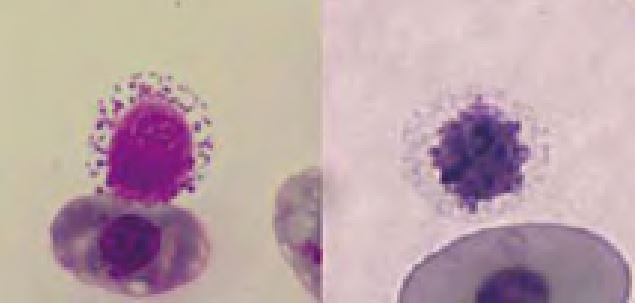
In some cases of regenerative anaemia, massive inflammation or post hibernation erythroid precursors may be found (Fig.4). These are not necessarily a sign of bone marrow disease as in mammals.
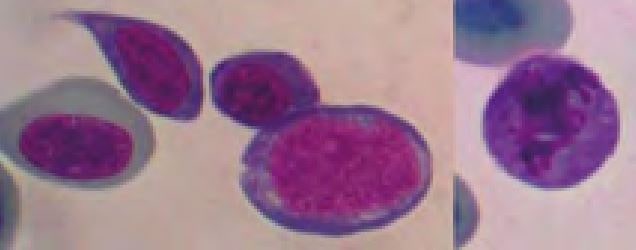
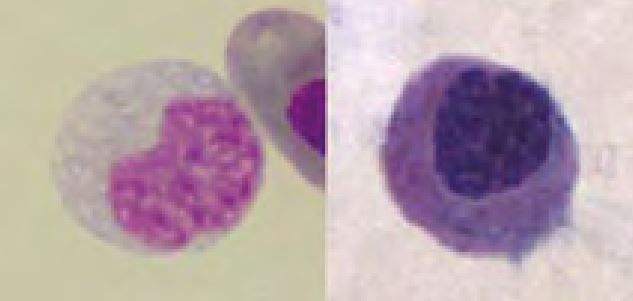
Thrombocytes are frequently aggregated on reptile blood smears which complicate the estimation of their number. However, if some aggregates (Fig. 5) are present the thrombocyte number is probably normal. The morphology of reptilian thrombocytes is variable, but they are always nucleated and mostly ellipsoid (Fig. 5). The cytoplasm is usually colourless, but can stain pale blue or pale violet. If the thrombocytes round up (e.g. because of activation) they can easily be confused with small lymphocytes (Fig. 6).
Reptilian leukocytes are classified as heterophils, eosinophils, basophils, lymphocytes, monocytes and azurophils. Classification is occasionally difficult, because the morphology of the blood cells sometimes varies significantly between reptilian species, as well as between subspecies of one species. The differential leukocyte count naturally exhibits a high variation, on the one hand between individuals due to season, temperature and partly gender, on the other hand due to species differences. Some reptile species show a lymphocyte pre-dominant haemogram, i.e. lymphocytes are the most prevalent cell (e.g. bearded dragon, iguana, tortoises), whereas in others granulocytes predominate.
Lymphocytes resemble those of mammals and are easily differentiated from other leukocytes. They have a round nucleus with dark chromatin and only a scant amount of basophilic cytoplasm.
Lymphocytosis occurs during inflammations and viral infections.
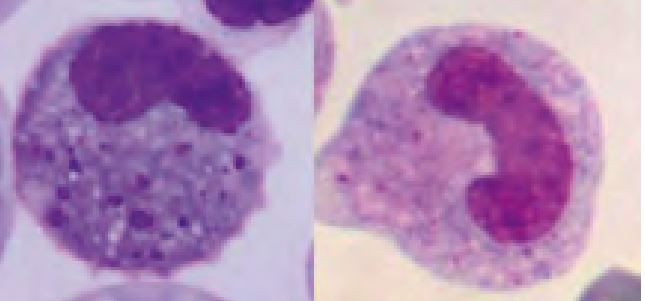
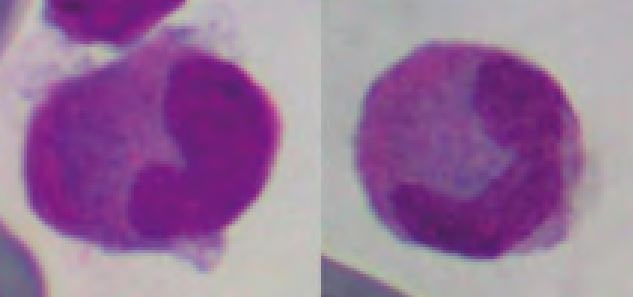
The function of heterophils is analogous to the function of neutrophils in mammals. Heterophilic granules stain orange to pink with quick stains. Sometimes granules dissolve and make the cytoplasm appear eosinophilic (Fig. 7, lower left). The differentiation of heterophilic and eosinophilic granules only by their colour is sometimes impossible. However, heterophils are mostly of larger size and the granules are long, while eosinophilic granules are almost always round.
The nucleus of heterophils is round in some species (e.g. tortoises, crocodiles, snakes); other species (e.g. iguanas, chameleons, dragons) show a segmented nucleus in mature cells.
Heterophilia is primarily associated with inflammation (often together with monocytosis). Other causes are stress, paraneoplastic syndrome or leukaemia (uncommon). In severe, acute inflammation animals that normally have segmented heterophils can show a left shift (bands). In extreme cases toxic changes are also visible: loss of physiologic granules, basophilic and/or vacuolated cytoplasm and toxic granulation (Fig. 8).
Eosinophils display bright red or orange, round granules in most reptiles. Some species (e.g. green iguana, bearded dragon) show also small and round, but blue to grey granules. The number of eosinophils varies highly between species (chelonians often show high numbers) and with various factors like season and parasitic infestation (Fig. 9).
Basophils have many dark purple staining granules that sometimes obscure the eccentrically placed nucleus. Using quick stains basophils often show only few granules or appear vacuolated (Fig. 10).
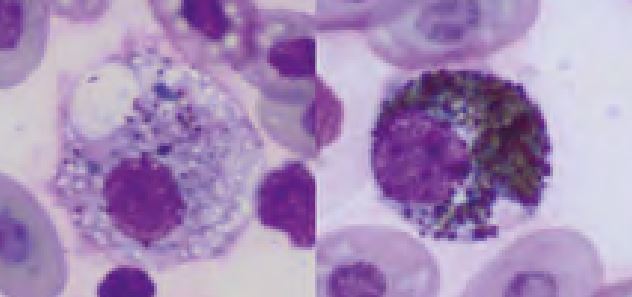
Monocytes have variably shaped nuclei, as in mammals. They have moderately blue-grey cytoplasm that can be clearly vacuolated in activated cells. Monocytosis is present in various inflammatory conditions, especially in granulomatous forms (Fig. 11).
Melanomacrophages are a special kind of monocytes in lower vertebrates that have phagocytized melanin granules. They can be found in inflammatory disease, especially in dermatitis (Fig. 12).
The origin of azurophils is an intensely debated topic. Morphologically the cells look like monocytes with dust-like eosinophilic granules in the cytoplasm (Fig. 13). Some authors classify them as subgroup of monocytes. Other investigators regard them as a separate cell type. In addition, the clinical significance of azurophils is still unclear.
- Fig. 1: Green iguana in it’s natural habitat
- Tab. 1: Sites for venipuncture in reptiles
- Fig. 2: Erythrocytes, thrombocyte (centre) and lymphocyte (right)
- Fig. 3: Left: normal erythrocytes and one thrombocyte, Right: erythrocytes displaying drying artefacts
- Fig. 4: Dysplastic erythroid precursors (left) and one erythrocyte in mitosis (right) of a chameleon with massive inflammation caused by a foreign body in the cloaca
- Fig. 5: Left: thrombocyte aggregate (Testudo hermanni), Right: diverse morphology of thrombocytes (upper: bearded dragon, lower: Boa constrictor)
- Fig. 6: left: small lymphocyte, right: thrombocyte
- Fig. 7: Heterophils: Upper left: Boa constrictor, Upper right: yellow-bellied slider, Lower left: marginated tortoise, Lower right: green iguana
- Fig. 8: Left: toxic band heterophil, Right: band heterophil (green iguana)
- Fig. 9: Eosinophils; Left: red-eared slider, Right: iguana
- Fig. 10: Basophils; the cell in the right image has no visible granules, the cytoplasm appears vacuolated
- Fig. 11: Monocytes; Left: red-eared slider, Right: Boa constrictor
- Fig. 12: Melanomacrophages, to the left with only few, to the right with many melanin granules in the blood of a green iguana with high grade dermatitis
- Fig. 13: Azurophils of a veiled chameleon