In Germany, vector-borne infections are becoming more important in dogs and cats for the following reasons:
- Import of dogs and cats from abroad.
- Increase in tourism and transport of goods in Europe and worldwide.
- Changes in climate.
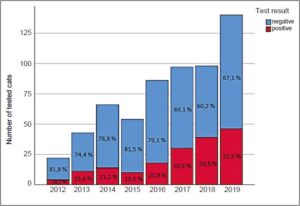
As a result of global warming, vectors which were previously endemic in warmer regions of the Mediterranean or in Southeastern Europe may be able to survive all year round in countries in Central and Northern Europe, such as Germany, and may even establish stable transmission cycles. Since April 2012, we have been offering a “Feline Travel Profile” for cats with suspected vector-borne infections related to a medical history of being abroad. This profile has been established in our range of services for cats that were imported into Germany from Mediterranean countries or Southeastern Europe or which have stayed in these countries as a travel companion (Fig. 1).
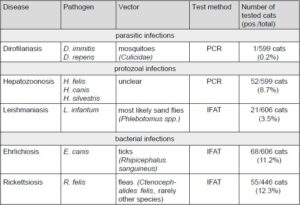
The “Feline Travel Profile” includes direct PCR detection of dirofilaria and hepatozoon and indirect antibody IFAT detection of leishmania, ehrlichia and rickettsia. More than a quarter of the cats (172/606; 28.4%) tested positive for at least one pathogen (Tab. 1).
Description of the tested pathogens
Hepatozoonosis
Hepatozoon (H.) felis is most frequently found in cats worldwide. In Europe, the pathogen has been detected in ticks (Rhipicephalus sanguineus, Ixodes hexagonus) and fleas (Ctenocephalides felis). The vector competence in cats is unclear. Transplacental infections with H. felis have also been described in cats. Recently, a first case report about an autochthonous infection with H. felis in a clinically affected domestic cat in Austria was published. Hepatozoonosis should, thus, also be considered in cats which have not had a history of staying abroad.
- Fig. 1: Number of cats tested between 04/2012 and 12/2019 using the “Feline Travel Profile” with the percentage of cats tested positive per year.
- Tab. 1: Vector-borne pathogens in the “Feline Travel Profile” with prevalence in the period 04/2012 – 12/2019.
- Fig. 2: Collected hard tick (symbolic image)
Leishmaniasis
In cats, Leishmania infantum is most likely transmitted by sand flies (Phlebotomus spp.). Dogs are considered the main reservoir, but cats can also become infected. However, it is assumed that cats are more resistant to infections than dogs. The pathogenesis of feline leishmaniasis is unclear, just like the role of the cat in the transmission cycle of the pathogen.
In cats, infections are often subclinical. When clinical signs occur, mainly nodular and ulcerative skin lesions are seen as well as enlarged lymph nodes, weight loss and ophthalmologic symptoms. To confirm infection, quantitative serological tests such as IFAT should be carried out in cats. In case titres are negative or low, a parasitological detection method (e.g. PCR, culture, histology, cytology) should be used to diagnose the pathogen if there is a clinical suspicion as seroconversion of the pathogen may be delayed for years.
Ehrlichiosis
In Europe, Ehrlichia (E.) canis is transmitted by the tick Rhipicephalus sanguineus. In Germany, the vector can only survive temporarily within certain temperature ranges or as populations in buildings heated throughout the year. Case studies on infections with E. canis in cats are rare and the clinical picture is non-specific. A single positive antibody detection by IFAT most likely indicates a pathogen contact in an endemic foreign country.
Rickettsiosis/Mediterranean spotted fever
More than 20 species belong to the “Spotted fever group”, with Rickettsia conorii being the most important zoonotic agent in Europe. Rickettsia conorii is transmitted by ticks (Rhipicephalus sanguineus) and can cause clinical signs in dogs; its significance in cats is not clear.
Rickettsia felis is transmitted by fleas. Seroconversion occurs in cats after contact with infected fleas. Bacteraemia is assumed to be short, as cats with confirmed antibodies usually had negative PCR results in blood. Antibody detection was often positive in cats in which several vector-borne infectious agents were found. This also reflects what is known about pathogen detection in dogs or in human medicine.
Dirofilariasis
Diroflaria (D.) immitis (heartworm) is a pathogenic species in cats, while D. repens, as the causative agent of subcutaneous dirofilariasis, also plays an important role as a zoonotic agent. Both pathogens are transmitted by mosquitoes. Prevalence in cats is only about one tenth of that in dogs. In cats, infections with D. immitis may be self-limiting, however, there may also be fatal courses with sudden death and clinical signs such as shortness of breath, coughing and vomiting. Yet, cats are more resistant than dogs and only occasionally develop transient microfilaraemia. Since larvae are only temporarily detectable in the blood and/or the number of larvae is below the detection limit of the D. immitis antigen detection test or the Knott’s test, false negative results often occur in cats. Heat treatment of the serum before performing the test increases the probability of pathogen detection.
We offer this method after prior consultation. Furthermore, a cardiac ultrasound should be performed to detect worms in the pulmonary artery as well as in the right atrium and the right ventricle of the heart. Diroflaria repens can cause skin lesions such as subcutaneous nodules.
Severe courses with itching, pustules, ulcerations and exfoliative dermatitis are rare. Microfilaria PCR has a low sensitivity in cats. If pathogen detection is positive, it should be followed by species differentiation to decide on the treatment (Fig. 2).
Summary
In cats with the appropriate medical history, vector contact and the respective clinical signs, vector-borne infections should also be considered in the differential diagnosis. Some vector-borne infections are probably underdiagnosed in cats. The role of the cat as a reservoir host and the clinical relevance of many vector-borne infections are not yet sufficiently understood. Preventive measures such as vector control throughout the year with suitable acaricidal and insecticidal preparations are also recommended for cats. The prepatent period of dirofilaria and leishmania is long. In case of negative test results within six months after import or return to Germany, it is thus recommended to repeat the test at the time when the cat has exclusively been in Germany for at least six months.