For sick guinea pigs, laboratory diagnostics remain an important part of the diagnostic workup. Due to the non-specific clinical signs, which for owners are only visible at a late stage, guinea pigs are often presented with rather advanced disease. At this point, laboratory diagnostics is usually crucial. It is also commended in cases of poor body condition, inappetence or as part of a preoperative screening in addition to further imaging techniques.
Pre-analytics
As guinea pigs and other small mammals are flight animals and therefore more susceptible to stress than dogs and cats, it is important to have all necessary materials ready before blood sampling, thereby reducing the manipulation time to a minimum.
Due to the small body size and total blood volume (69-75 ml/kg), small blood tubes are recommended. Lithium heparin tubes are particularly suitable for guinea pigs, as it is possible to determine T4 and fT4, besides analysing clinical-chemical parameters and a complete blood count. Lithium heparin blood (1 ml) is therefore a good alternative to EDTA blood and serum if, one expects a small amounts of blood upon sampling.
Furthermore, it is recommended to make blood smears directly in the practice in order for the lab to be able to evaluate the differential blood count in case of transport-related cell degenerations.
Puncture sites
Blood can be collected at different puncture sites using a G20 – G21 needle (with or without cone).
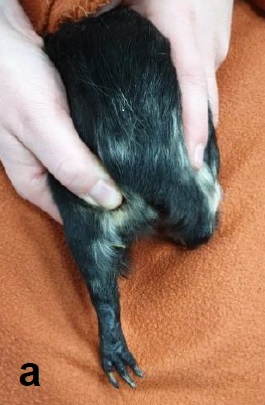
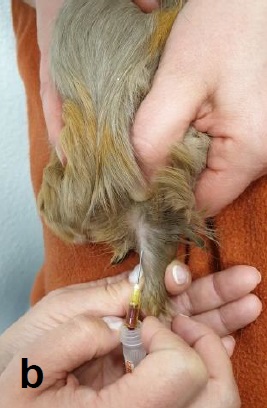
The lateral saphenous vein is most suitable for this. It is located caudally on the lower leg and is clearly visible and palpable (Fig. 1a). A puncture (Fig. 1b) is performed at half height between the femoral canal and the tarsal joint at an angle of 30° to the leg axis.
It is also possible to puncture the vena Cephalica antebrachii, located laterally on the foreleg (Fig. 2). Here, it is recommended to position the leg parallel to the edge of the table.
Haematology
Compared to other species, guinea pigs have fewer, but larger erythrocytes (6.6- 7.9 µm). Haematocrit (0.39-0.55 l/l) is similar to that of dogs; young animal anaemia can also occur. Determination of reticulocytes for the classification of anaemia is also possible in guinea pigs (Kaufhold et al., 2017).
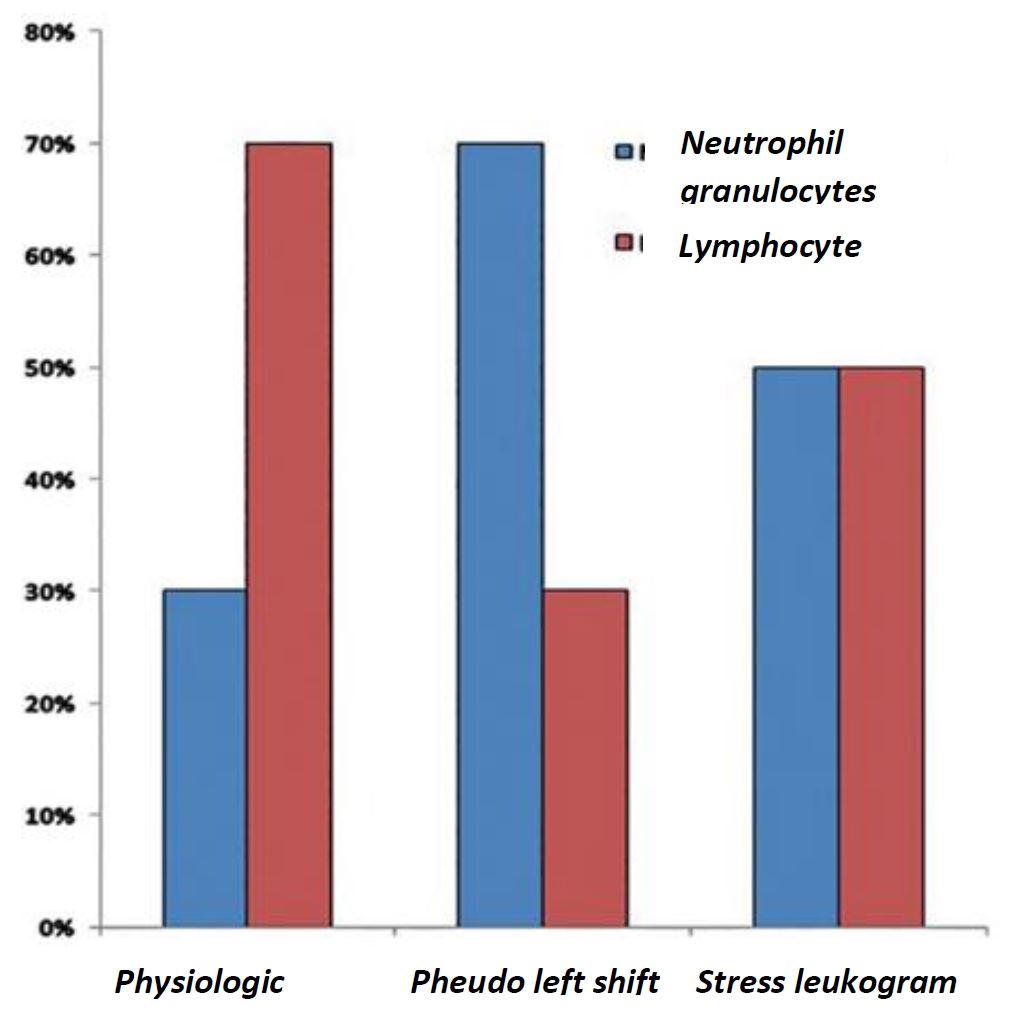
In contrast to dogs and cats, guinea pigs have a lymphocytic blood count. Physiologically, more lymphocytes than neutrophil granulocytes circulate in the peripheral blood. By acute bacterial infections or inflammation, a so-called “pseudo left shift” occurs – a shift from lymphocytic to granulocytic blood count. In this case, a marked leukocytosis as well as the presence of band neutrophils are, compared to other species, rarely seen. By the so-called “stress leukogram” are lymphocytes and granulocytes almost equally distributed.
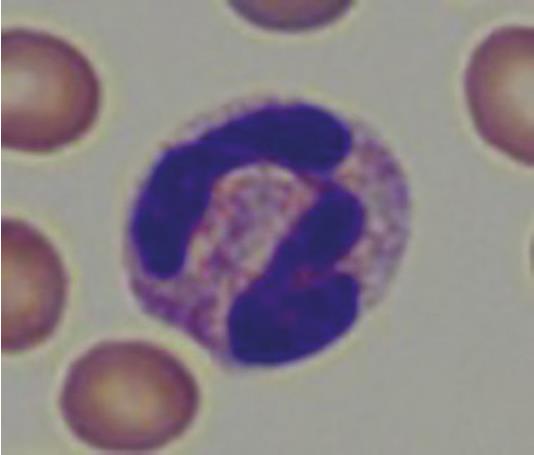
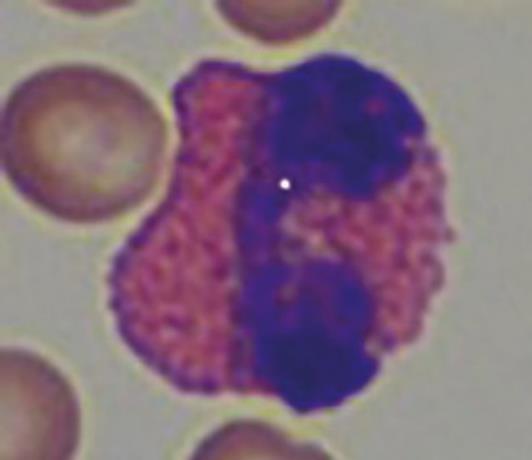
Neutrophil granulocytes in guinea pigs are also referred to as “pseudo-eosinophils” (Fig. 4) due to their acidophilic staining. These can easily be confused with eosinophil granulocytes, but are smaller in comparison, as their nucleus is less segmented and contains smaller granules.
Eosinophilia occurs, in contrast to rabbits, not only by tissue injury and infections of tissue containing mastocytes (skin, lungs, gastrointestinal tract, uterus), but also by parasitosis.
Guinea pigs can also develop leukaemic lymphomas with severe leukocytosis, which, compared with other species, can easily be diagnosed by a blood examination.
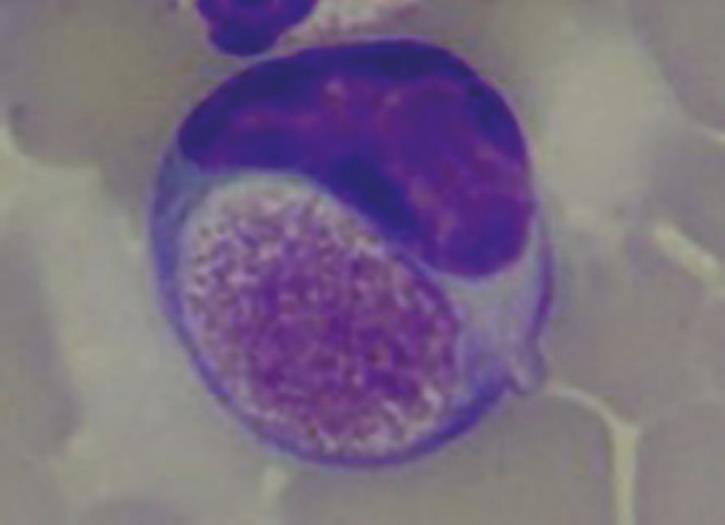
Foa-Kurloff cells (Fig. 6) represent one last haematological feature of this species. Foa-Kurloff cells are mononuclear leukocytes (often lymphocytes, rarely monocytes) with inclusion bodies (Kurloff bodies), which should not be mistaken for intracellular pathogens, due to their appearance. They can make up 3-4% of the leukocytes.
A particularly high number of these inclusion bodies is described during pregnancy and correlates with the oestrogen level. They are most likely equivalent to the killer cells in other mammals and provide a barrier between foetus and mother.
Clinical chemistry
Most laboratories offer so-called Rodent Profiles, which provide a good overview of the individual metabolic status. Often liver, kidney and muscle parameters included, as well as fructosamine, total protein and electrolytes.
Most laboratories offer so-called Rodent Profiles, which provide a good overview of the individual metabolic status. Often liver, kidney and muscle parameters included, as well as fructosamine, total protein and electrolytes.
In order to assess hepatic metabolism, attention should be paid to enzyme changes in GLDH, ALT and AST. In guinea pigs, GLDH is a sensitive enzyme and increases rapidly by acute hepatopathies, although it can be found in kidney cells too. As ALT and AST occur in low concentrations, not only in the liver, but also in other organs, they are not specific for hepatocellular damage. Increased enzyme concentrations, combined with other elevated liver parameters, indicate chronic hepatic disease. Increased AST activity should always be assessed in conjunction with CK, as AST is not only found in the liver, kidney and pancreas, but also in myocardial and skeletal muscle cells.
Although the lipid metabolism in guinea pigs is less active than that of rabbits, they tend to develop hepatic lipidosis. Both AP and γ-GT are slow to react and only increase by chronic conditions. Evaluation of urea and creatinine does not differ from other animal species.
As especially coprophagic herbivores never fast (and should not be fasted before blood sampling), they generally have high glucose levels of 5.0-16.0 mmol/l. However, the physiological fructosamine level is comparatively low at < 271 µmol/l. Glucose is a significant prognostic factor by ileus or stasis. The higher and longer a hyperglycaemia persist, the worse the prognosis for the affected animal. An additional drop in sodium (Na) (< 129 mmol/l) increases the mortality rate by a factor of 2.3 (Bonhevi et al., 2014).
Endocrinology
In contrast to dogs and cats, hormonal diseases in guinea pigs are described in low number. Cases of hyperthyroidism, Morbus Cushing, diabetes mellitus and insulinoma are reported.
Hyperthyroidism
Hyperthyroid animals are often presented at an older age, with progressive weight loss, despite a constant or increased appetite, proliferation of the ventral neck as well as behavioural changes. As the disease progresses, PU/PD, alopecia, inappetence, chronic diarrhoea and cardiac problems can occur.
For diagnosis, T4 should be determined (at least 250 µl of serum or heparin plasma). To exclude differential diagnoses, blood, urine and faecal examinations are recommended as well as a cytological examination of the mass.
Morbus Cushing / Hyperadrenocorticism
Patients with suspected Morbus Cushing are often presented with bilateral alopecia beginning on the flanks and the ventral abdomen, apathy, PU/PD, polyphagia and bilateral exophthalmos. Thinner skin, weight loss and muscular atrophy may also be seen later in the course of the disease.
Diagnostically, a dexamethasone suppression test, as well as an ACTH stimulation test (from blood or saliva) can be used. Due to the pulsatile secretion of ACTH at a high frequency, guinea pigs generally have high baseline cortisol levels in their blood. Any stress, such as blood sampling, additionally increases the cortisol level.
A low-stress alternative is to measure the cortisol level from saliva. The ACTH stimulation test procedure is the same using saliva as from blood. A baseline sample is taken, 20 I.E. ACTH is injected i.m., and the stimulation sample is taken four hours post injection. It is mandatory to use Salivettes for obtaining saliva samples (Fig. 7, Salivette®, Sarstedt).
- Fig. 1: (a) Vena Saphena lateralis caudally on the lower limb
- Fig. 1: (b) Puncture of vena Saphena lateralis with a G20 needle
- Fig. 2: Vena Cephalica antebrachii laterally on the foreleg
- Fig. 3: Distribution of leukocytes
- Fig. 4: “Pseudo-eosinophil”
- Fig. 5: Eosinophil granulocyte
- Fig. 6: Foa-Kurloff cell with inclusion body of up to 8 µm in size
- Fig. 7: Salivette
- Fig. 8: Saliva sampling using a Salivette®
The plastic fibre roll inside the Salivette® is placed between the molars and buccal mucosa for five minutes. To prevent it from sliding, the plastic fibre roll can be fixated (Fig. 8). After successful sampling, the plastic fibre roll is placed back into the Salivette® and shipped to the lab.
A Dexamethasone suppression test (LDDS with 0.01 mg/kg or HDDS with 0.1 mg/kg of dexamethasone) is possible, although, no valid reference values are available so far in the literature. Similar to dogs, the suppression of the cortisol level is decisive in this case.
However, in guinea pigs Morbus Cushing is still a diagnosis of exclusion and should be verified by further examinations, such as sonography of the adrenal glands.
Diabetes mellitus
Affected animals mainly suffer from obesity and PU/PD. Bilateral cataracts are described. Both, the hereditary type I, as well as the acquired type II occur.
Laboratory diagnostics show a marked hyperglycaemia, increased fructosamine levels and glucosuria. To exclude differential diagnoses, a Rodent Profile including complete blood count, T4 and an ACTH stimulation test, as well as a urinalysis should be performed.
Insulinoma
So far, only two cases are described in the literature. Weakness, paralysis as well as convulsions and seizures were described. Low glucose and fructosamine levels were found on the clinical-chemistry analysis. Insulin measurement is not recommend-ed, due to intermittent secretion and lack of reference values.
Conclusion
Blood testing in guinea pigs has established itself over the past 50 years, due to the steadily growing number of domestic small mammals. However, further research is needed.