Platelets
Platelets, also called thrombocytes, are an important component of blood coagulation. They are anucleate, flat, irregularly round, oval or elongated cells and have light blue cytoplasm, which may contain fine, azurophilic granules.1 So-called “pseudopodia” may be visible in activated platelets.
Platelets are formed in the bone marrow. Megakaryocytes differentiate from bipotent megakaryocyte-erythrocyte precursors. By fragmentation, macrothrombocytes (Fig. 1) and platelets are formed.2
Depending on the literature, the average survival time of platelets in the peripheral blood is indicated with 3 – 9 days.1
Compared to other mammals, horses have one of the lowest platelet counts.1 In the blood smear, 6 – 10 platelets per visual field (1000x magnification with immersion oil) are sufficient for adequate platelet numbers. Depending on the literature, the reference range varies between 90 – 350 G/l (Laboklin 90 – 300 G/l).1
Pre-analytics
Pre-analytics plays an important role in the reliability of laboratory diagnostic findings. This, however, applies to all haematological parameters, not only to platelets. Improper blood collection, too much pressure or a wrong sequence when filling the tubes can lead to an activation of the platelets. This often leads to smaller or larger clots which can falsify the analysis or even make it impossible. Blood collection is best carried out in stress-free conditions and not after exertion (training). Excessive pressure should be avoided. Concerning the order of the tubes, the serum tube should ideally be filled first and the EDTA tube last. If only EDTA blood is collected, the first drops of blood should be discarded.
In horses, platelet count is quite independent of storage conditions for up to 24 hours after blood collection.3
Platelet count must be performed from anticoagulated whole blood. When comparing the anticoagulants K3-ethylenediaminetetraacetic acid (EDTA), lithium heparin (heparin) and sodium citrate (citrate), significant deviations regarding the platelet parameters can be detected in heparin blood.3 The use of heparin as an anticoagulant resulted in increased formation of platelet aggregates and, thus, falsely low platelet levels were measured.4 EDTA is the most suitable anticoagulant for blood analysis.4 However, EDTA-induced pseudothrombocytopenia has also been described in individual cases in horses, leading to increased formation of platelet aggregates.7 A reduced number of aggregates can be achieved by measuring quickly within 2 hours after blood collection and by warming the sample to 37 °C before measurement.8
EDTA is the anticoagulant of choice for determining platelet count. The blood should be analysed within 24 hours and stored cooled, if possible.
Tip: Place the blood samples in a polystyrene box in your medical practice car to protect them from heat and frost!
-
Fig. 1: Macrothrombocytes, 1000x magnification, immersion oil, Giemsa stain
Picture credits: Laboklin
-
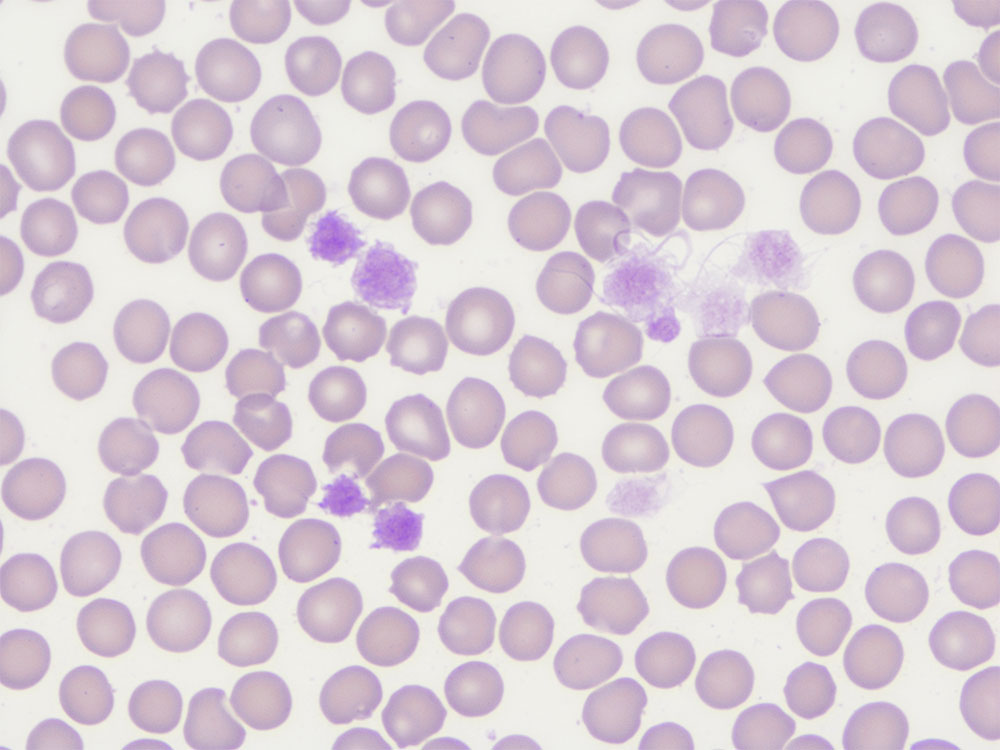
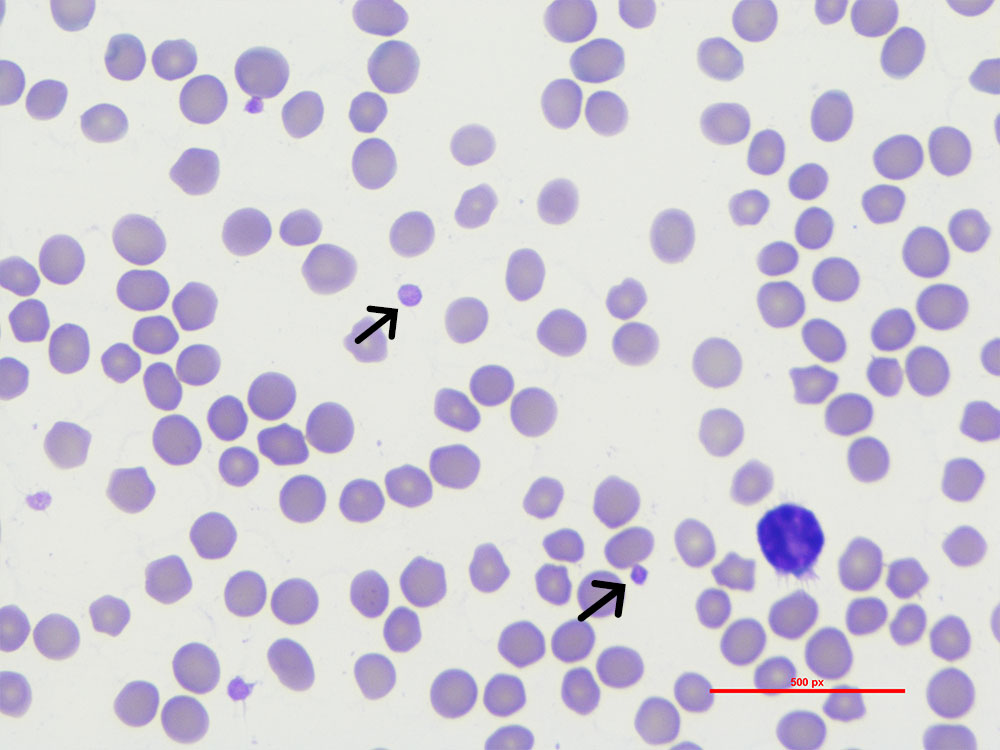
Fig. 2a: Erythrocytes and platelets (black arrow), Giemsa stain, 1000x magnification, immersion oil
Picture credits: Laboklin
-
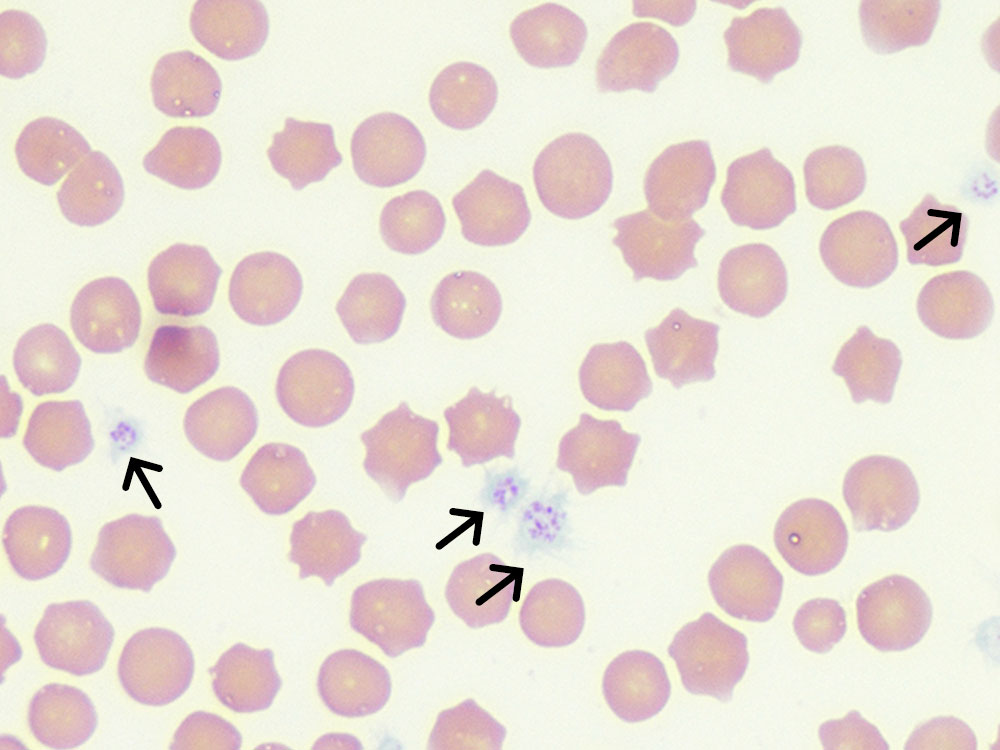
Fig. 2b: Erythrocytes and platelets (black arrow), Diff-Quik stain, 1000x magnification, immersion oil
Picture credits: Laboklin
-
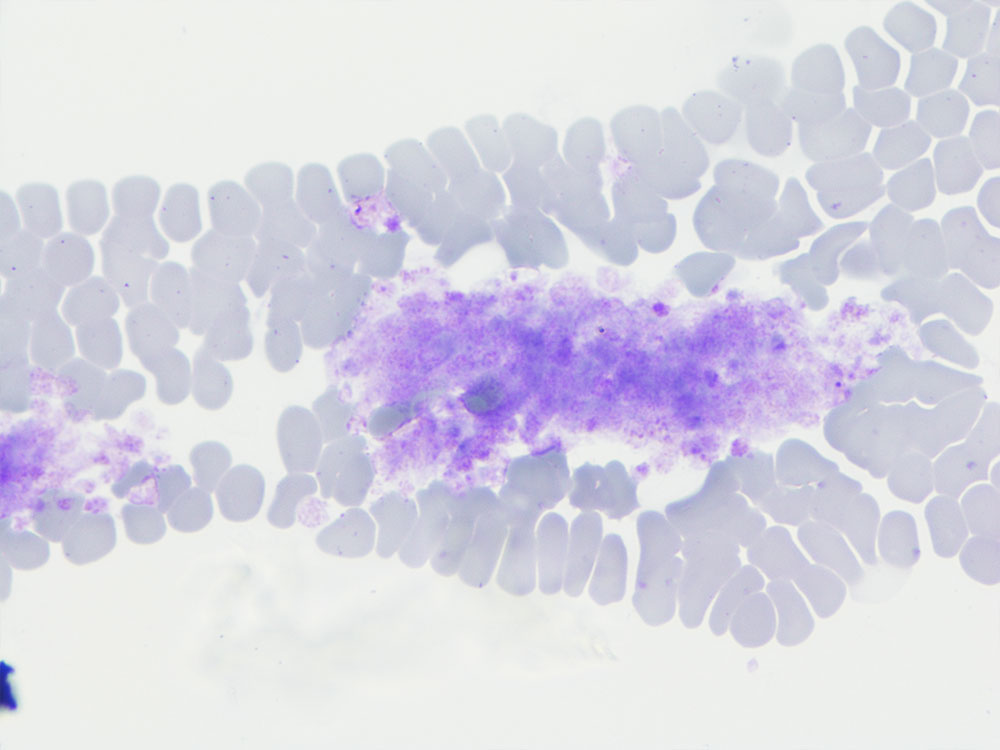
Fig. 3: Large platelet aggregates in the feathered edge of the blood smear. The number of platelets in the aggregates cannot be determined. 100x magnification, Diff-Quik
Picture credits: Laboklin
Laboratory diagnostics
Platelet counts are usually included in every complete blood count. They can, however, also be requested individually. Additionally, you should also always prepare a blood smear directly before or after platelet analysis. This blood smear can be used to look for aggregates when platelet counts are low or to confirm thrombocytopenia. To evaluate the blood smear, it is air-dried and then stained. You can, for example, use Wright-Giemsa or Diff-Quik for staining (Figures 2a and 2b).
For further evaluation, a detailed clinical history (e.g. contact with viral, bacterial, parasitic infectious agents? Fever? Gastrointestinal signs? Respiratory signs?) and a thorough examination of the patient are necessary.
Microscopic examination of the platelets is first carried out at 100x magnification (10x objective). The feathered edge is examined for platelet aggregates. Platelets are then counted in the monolayer at 1000x magnification (100x objective). In order to calculate the approximate platelet count, the average of 10 visual fields is multiplied by 15 – 20. Any aggregates in the feathered edge must also be taken into account. This blood smear can be used to look for aggregates (Fig. 3) when platelet counts are low or to confirm thrombocytopenia. For example: In the monolayer, 98 platelets are counted in 10 visual fields. The average is 9.8 platelets per visual field. The number is rounded up to 10 and then multiplied by 15.
Thus, the estimated platelet count of the horse is 150 G/l. Rough estimates of any aggregates must be added to this figure.
If thrombocytopenia is confirmed by the blood smear, further examinations are recommended depending on the clinical history and the suspected diagnosis, e.g.
-
- PCR tests: e.g. Babesia, Anaplasma
- coagulation tests: PT, aPTT, thrombin time, fibrinogen
- serological tests: e.g. equine infectious anaemia (EIA), equine herpesvirus (EHV)
- haematology: antiplatelet antibodies
- bone marrow biopsy and cytology, if necessary
Thrombocytopenia
In most cases, thrombocytopenia is an incidental finding in horses. Thrombocytopenia is a decrease in platelet numbers below the species-specific norm.9 Thrombocytopenia usually indicates a pathological process which can lead to coagulation disorders when platelet count is significantly reduced.10 There is often more than one cause for thrombocytopenia.12
A distinction must be made between pseudothrombocytopaenia and true thrombocytopenia. Pseudothrombocytopenia occurs if not all the platelets present are counted in the platelet count. Manual microscopic examination is therefore always recommended as a first control mechanism in thrombocytopenia.10
Reasons for true thrombocytopaenia are reduced production in the bone marrow, increased consumption, destruction of platelets or increased sequestration (Table 1).10, 11
Table 1: Summary of causes of thrombocytopenia 10, 11
| Cause | ||
| Thrombocytopenia | pseudothrombocytopenia | limitations of measurement
|
| reduced production in the bone marrow |
|
|
| increased consumption |
|
|
| destruction of platelets | immune-mediated destruction
non-immune-mediated destruction
|
|
| sequestration |
|
|
Clinical signs of thrombocytopenia
In most horses, thrombocytopenia occurs secondary to a variety of underlying diseases.12
The clinical picture usually corresponds to the causative disease. In general, signs are rare in low to moderate thrombocytopenia (90 – 30 G/l) as long as secondary haemostasis is not impaired.
Severe thrombocytopenia (< 30 G/l) leads to changes in primary haemostasis.13, 14 Patients show petechiae and ecchymoses, which are mainly seen on the mucous membranes and the sclerae. Increased epistaxis has also been described. There is prolonged bleeding after blood sampling.
Thrombocytosis
Thrombocytosis is defined as an increase in the number of platelets circulating in the peripheral blood above the species-specific reference range. As with thrombocytopenia, a distinction can be made between pseudothrombocytosis and true thrombocytosis (Table 2). Pseudothrombocytosis occurs when other cells are counted as platelets in the platelet count. This can include small erythrocytes or lysed erythrocytes. The evaluation of scatterplots and blood smears helps here.
Mild, physiological thrombocytosis often occurs during and after exercise or agitation (e.g. stress during blood collection). This is caused by splenic contractions which release stored platelets into the circulation.18, 19
True thrombocytosis can be classified into primary and secondary thrombocytosis. In most cases, platelet count increase in secondary thrombocytosis is only mild to moderate (300 – 500 G/l), whereas it is high in primary thrombocytosis. Furthermore, primary thrombocytosis is more frequently associated with clinical bleeding and thrombosis.20, 21, 22
Secondary thrombocytosis mostly occurs in inflammation and infections.24
Table 2: Summary of causes of thrombocytosis 11, 22, 23
| Cause | ||
| Thrombocytopenia | pseudothrombocytosis | limitations of measurement
|
| redistribution of platelets |
|
|
| increased production in the bone marrow |
|
|
| neoplasia |
|
|
Conclusion
Platelet counts are part of almost every blood count and may provide an indication of an underlying disease. In any case, their plausibility must first be checked manually under the microscope. The cause of thrombocytopenia or thrombocytosis must then be found based on a good medical history and clinical examination.
Dr. med. vet. Annemarie E. Baur-Kaufhold